New strains to provide crucial insights in mental health research across the sector
VANCOUVER, British Columbia, May 17, 2022 (GLOBE NEWSWIRE) --
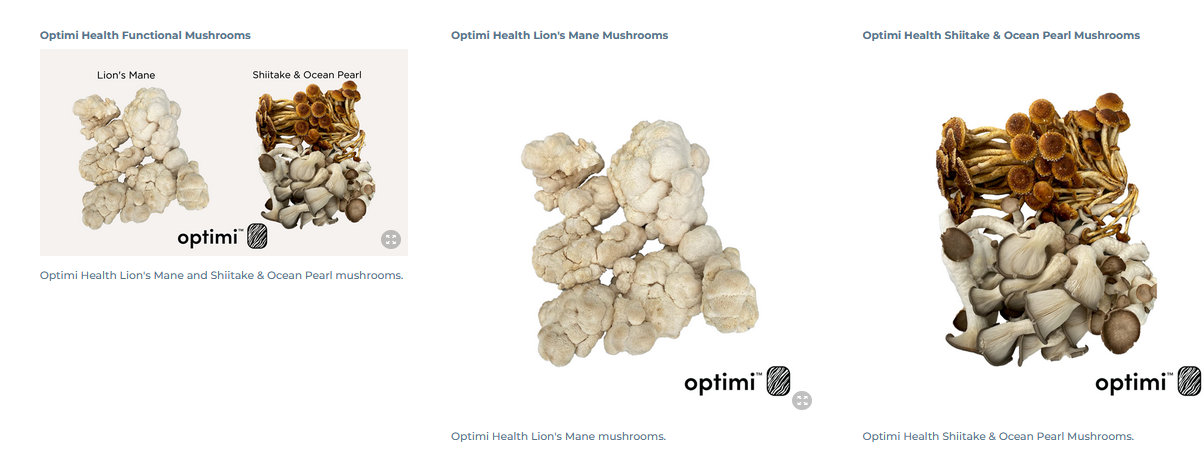
Optimi Health Corp. (CSE: OPTI) (OTCQX: OPTHF) (FRA: 8BN) (“Optimi” or the “Company”), a homegrown, Canadian company producing natural, scalable, and accessible mushroom formulations for transformational human experiences, is pleased to announce that it has completed the acquisition of a diverse catalog of psilocybin and functional mushroom strains intended for cultivation in its 20,000 square foot, EU-GMP compliant facility in Princeton, British Columbia.
The Company’s acquisition includes 24 psychedelic and nine functional strains, giving it one of the largest genetic banks in the sector. Formulated psychedelic strains will be made available to licensed researchers, Canada’s Special Access Program (SAP), as well as being utilized for on-site research in Optimi’s recently expanded analytical laboratory, while the functional strains will be optimized for the Company’s growing whole body, natural supplement brand,
Optimi Life.
Last week, Optimi
applauded Health Canada’s
Notice to Stakeholders which specified the “importance of drug quality and Good Manufacturing Practices (GMP)” in supplying psilocybin to clinical trials and the SAP. As a leading cultivator of natural EU-GMP psilocybin, Optimi will begin processing its genetics for supply to patients requesting natural product.
In keeping with its overarching goal of the scalable production of safe, EU-GMP psilocybin, the Company is undertaking the establishment of an industry-leading base of mycological genetics with the aim of determining the comparative efficacy of various strains of psychedelic mushrooms. Further, this acquisition will strengthen Optimi’s position as a tier-one ingredient supplier of naturally occurring psilocybin to approved entities engaging in molecular development and clinical trials.
"We want to be in a place where we can facilitate as broad a spectrum of research as possible,” said Optimi Health Chief Science Officer Justin Kirkland. “From the data available so far, we can say with relative certainty that psychedelic research and any novel psilocybin-assisted therapies that may result are not going to be a one-size-fits-all process. It is entirely possible that certain strains of mushrooms with varying chemical compositions and potency will lead to varying degrees of efficacy across a wide array of mental health indications when paired with the appropriate therapeutic protocols.”
By collating information from clinical trials, using mushrooms from both its own supply and other sources throughout the sector, Optimi is committed to working within the regulatory approval processes necessary to ensure access to those in need.
Optimi CEO Bill Ciprick expanded on the shared scientific and commercial advantages provided by the company’s comprehensive knowledge of psilocybin genetics.
“In conjunction with the recent completion of our analytical laboratory, this acquisition is intended to position Optimi as the leading supplier of naturally-sourced psilocybin to the burgeoning psychedelic industry,” said Ciprick. “Optimi’s strategic year of commercialization is based around our growing capacity to supply and analyze as many strains of GMP psilocybin as may be necessary to ensure the success of the hundreds of clinical trials and drug discovery efforts currently underway, underpinning both the future of psychedelic science and the commercial viability of the space as a whole,” Ciprick concluded.
GRAND OPENING
On May 27th, the Company will be welcoming guests and dignitaries from the psychedelic business space, the medical and public health fields, local and national politics, and the Indigenous community for a grand opening event and facility tour to inaugurate its Princeton, BC facility. For those interested in attending in-person or via livestream should contact event organizer, Michael Kydd, at
info@kyddergroup.com.
ABOUT OPTIMI (CSE: OPTI) (OTCQX: OPTHF) (FRA: 8BN)
Optimi Health is a homegrown, Canadian success story producing and supplying natural, EU-GMP grade psilocybin and functional mushrooms that focus on the health and wellness markets. Built with the purpose to produce scalable, natural mushroom formulations for transformational human experiences, the Company’s goal is to be the number one trusted, compassionate supplier of safe, natural GMP grade psilocybin across the world. With a vertically integrated approach, Optimi intends to cultivate, extract, process and distribute high quality functional and psychedelic mushroom products at its two facilities comprising a total of 20,000 square feet in Princeton, British Columbia.
To fully investigate the science of mushrooms, the Company has received a research exemption under Health Canada Food and Drug Regulations (FDR) for the use of Psilocybin and Psilocin for scientific purposes via its wholly owned subsidiary Optimi Labs Inc. Optimi has also been granted a dealer’s license under Canada’s Narcotic Control Regulations governing possession, distribution, sale, laboratory analysis of and research and development of Psilocybin and Psilocin formulations. Optimi is committed to expert cultivation and quality production subject to and in accordance with the terms of all applicable laws and governing regulations to ensure safe, superior Canadian fungi production.
FOR INTERVIEW REQUESTS OR FURTHER INFORMATION PLEASE CONTACT:
Investor Relations
Email: investors@optimihealth.ca
Phone: +1 (902) 880 6121
Web: https://optimihealth.ca
FORWARD‐LOOKING STATEMENTS
This news release contains forward‐looking statements and forward‐looking information within the meaning of Canadian securities legislation (collectively, “forward‐looking statements”) that relate to Optimi’s current expectations and views of future events. Any statements that express, or involve discussions as to, expectations, beliefs, plans, objectives, assumptions or future events or performance (often, but not always, through the use of words or phrases such as “will likely result,” “are expected to,” “expects,” “will continue,” “is anticipated,” “anticipates,” “believes,” “estimated,” “intends,” “plans,” “forecast,” “projection,” “strategy,” “objective,” and “outlook”) are not historical facts and may be forward‐looking statements and may involve estimates, assumptions and uncertainties which could cause actual results or outcomes to differ materially from those expressed in such forward‐looking statements. No assurance can be given that these expectations will prove to be correct and such forward‐looking statements included in this news release should not be unduly relied upon. These statements speak only as of the date of this news release. In particular and without limitation, this news release contains forward‐ looking statements pertaining to activities proposed to be conducted under the Company’s dealer’s license and associated business related to Psilocybin and Psilocin and Optimi’s plans, focus and objectives.
Forward‐looking statements are based on a number of assumptions and are subject to a number of risks and uncertainties, many of which are beyond Optimi’s control, which could cause actual results and events to differ materially from those that are disclosed in or implied by such forward‐looking statements. Such risks and uncertainties include, but are not limited to, the impact and progression of the COVID‐19 pandemic and other factors set forth under “Forward‐Looking Statements” and “Risk Factors” in the Company’s Annual information Form dated January 12, 2022, and other continuous disclosure filings available under Optimi’s profile at www.sedar.com. Optimi undertakes no obligation to update or revise any forward‐looking statements, whether as a result of new information, future events or otherwise, except as may be required by law. New factors emerge from time to time, and it is not possible for Optimi to predict all of them or assess the impact of each such factor or the extent to which any factor, or combination of factors, may cause results to differ materially from those contained in any forward‐looking statement.
Any forward‐looking statements contained in this news release are expressly qualified in their entirety by this cautionary statement.
Photos accompanying this announcement are available at:
https://www.globenewswire.com/NewsRoom/AttachmentNg/85ea3395-7e18-4ad2-8362-d18637d52e31
https://www.globenewswire.com/NewsRoom/AttachmentNg/61a31bf5-5b93-4b99-9ca6-e566bfc40195
https://www.globenewswire.com/NewsRoom/AttachmentNg/1d9cae60-a9d7-4885-ab01-06496bf7d702